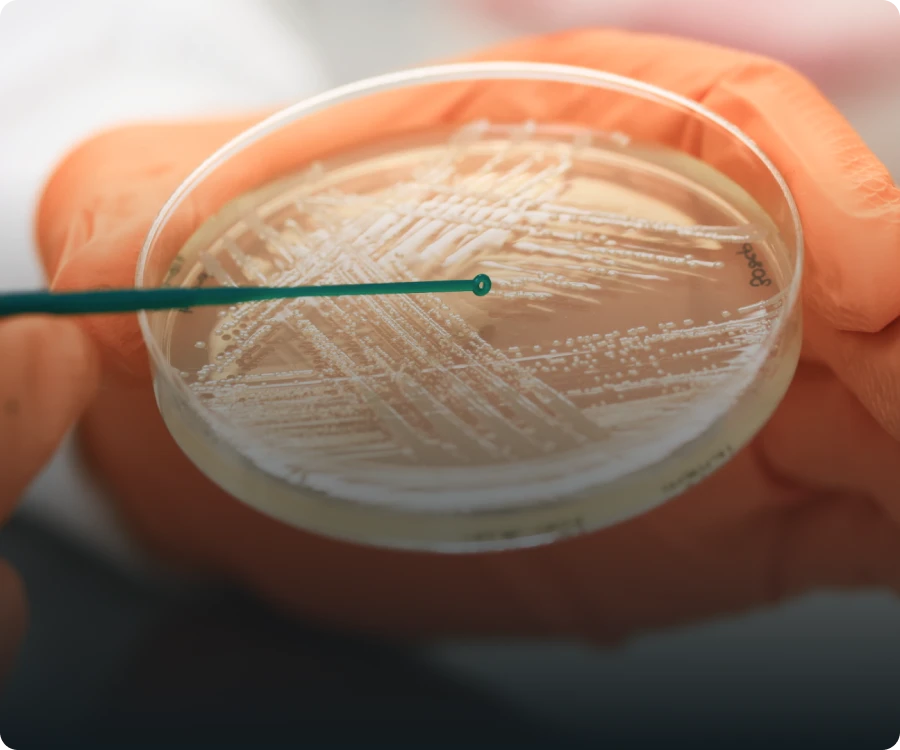
Worlds biggest
manufacturers
of bacteriophages
PARTNER WITH US
MORE THAN 10 YEARS OF SUCCESSFUL TRACK RECORD
PARTNER WITH US

PRODUCED MORE THAN
150
Cell/Viral Banks

PRODUCED MORE THAN
120
DSs/APIs

PRODUCED MORE THAN
14
DPs for Clinical Trials

DEVELOPED MORE THAN
200
Specific methods

QUALIFIED MORE THAN
500
Methods

RUNNING MORE THAN
150
Stability programs
OUR GLOBAL NETWORK
.svg)

Fast turnaround time
We prioritize efficiency to ensure your projects are completed on time, every time.
CLEAR & DIRECT COMMUNICATION
Our team keeps you informed at every stage with open and straightforward communication.
PRECISE TIMELINE COORDINATION
We align every step of the process to meet your deadlines with accuracy and reliability.
COST EFFECTIVE SOLUTION
Our services are designed to deliver maximum value without compromising on quality.
SCALABILITY & EXPERTISE IN GMP PRODUCTION
From small batches to large-scale manufacturing, we offer GMP-compliant solutions tailored to your needs.
CONNECT WITH AN EXPERT
CONTRACT RESEARCH & MANUFACTURING PARTNER




JUST RELEASED
.png)